How Many Degrees Is a Bent Molecule
Certain atoms such as oxygen will almost always set their two or more covalent bonds in non-collinear directions due to their electron configuration. The molecule is two dimensional and bent as opposed to the beryllium.
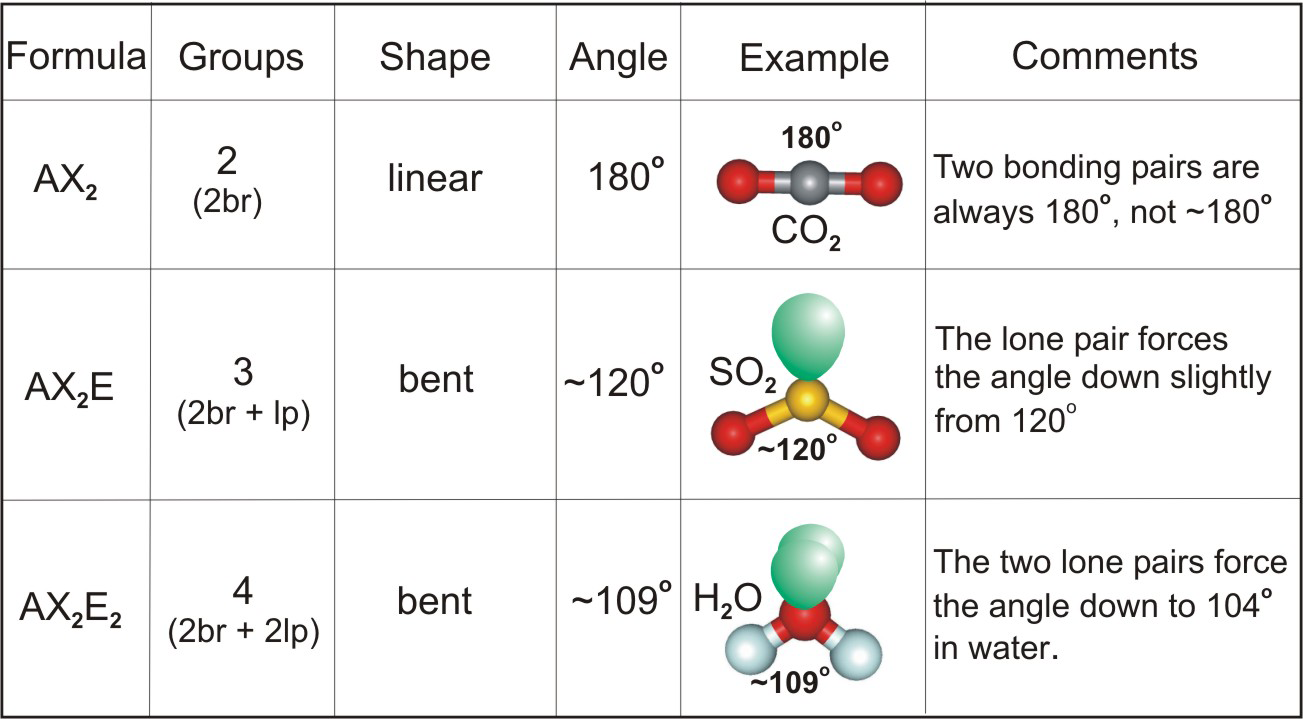
The most common actual angles are 105 107 and 109.

. They have central angles from 104 to 1095 where the latter is consistent with a simplistic theory which predicts the tetrahedral symmetry of four sp3hybridised orbitals. Water is the most commonly known bent molecule. In a bent molecule the angle between two of the atoms is approximately 10445 degrees.
For bent molecular geometry when the electron-pair geometry is tetrahedral the bond angle is around 105 degrees. On the other hand. What are Linear Molecules.
Hence it has 3 translational and 3 rotational degrees of freedom. See the answer See the answer See the answer done loading. Here are some examples of the Bent shape molecule.
Also Know why is a molecule bent. Beside above what is the degree of a bent molecule. The water molecule is bent molecular geometry because the lone electron pairs although still exerting influence on the shape are invisible when looking at molecular geometry.
In chemistry molecules with a non-collinear arrangement of two adjacent bonds have bent molecular geometry also known as angular or V-shaped. This is why the water molecule is a bent shape rather than a linear shape - it has two lone pairs of electrons giving it. This bend provides the bond angle of less than 1095 degrees 1044 degrees.
The repulsion caused by the addition of these extra electrons causes the molecule to become bent. To a 104obond angle. For bent molecular geometry when the electron-pair geometry is tetrahedral the bond angle is around 105 degrees.
The angle of bent molecules is less than 120 if there is one pair of nonbonding electrons and is less than 1095 if there are two pairs of nonbonding electrons. How many degrees is a bent molecule. In the case of bent molecules that shape is always between 1095 and 104 degrees.
In the case of water although it has two bonds that are connected to the H it also has two lone pairs which push down on the two bonds and make a bent shape rather than it being linear. NO2 is a bent molecule. If the molecule has 3 regions of electron density but one is a lone pair then it can be bent 120 degrees.
How many vibrational degrees of freedom does water have H2O is a bent molecule Question. However when you remove an electron from it making it NO2 the molecule becomes linear due to the loss of a lone electron. A A molecule with N atoms has 3N degree of freedoms.
However if the molecule has 4 regions of electron density and two of them are lone pairs then it can also be bent 1095 degrees. Postby LilianKhosravi_1H Mon Jun 04 2018 113 am. Moreover linear molecules are straight molecules having a bond angle of 180 degrees while bent molecules are angular molecules having a bond angle that is below 180 degrees.
Up to 24 cash back The molecule is polar since it is asymmterical and bond angles are less than 1044 degrees. This repulsion of the lone pairs of electrons on the oxygen atom causes the bond of the hydrogen to. Bent molecules occur most frequently in group 16 elements because they have 6 electrons in their outer shell allowing for 2 bonds and 2 lone pairs.
They vary because of the different properties of the peripheral atoms X. Hence since our moleculeSO2 has 3 atoms the total number of degrees of freedom is 9. You would know the molecule is bent if it has any lone pairs.
The bond angle bet. The rem View the full answer. How many vibrational degrees of freedom does water have H2O is a bent molecule This problem has been solved.
The reason water has a bent shape is that the two lone pair of electrons are on the same side of the molecule. B SO2 is a non-linear molecule. Water H2O is an example of a bent molecule as well as its analogues.

As Studypeach Shapes Of Molecules

Comments
Post a Comment